Why Does Modern Pharmacology Use Pure Chemicals to Make Many Prescription Drugs?
ID 261391534 | Formula Thc © Allexxandar | Dreamstime.com
Recently I have been writing about the Trump Administration’s proclivity to shape drug policy by Executive Order. There is more to say about the subject.
In the late 19th century, drugs began to be developed using pure chemicals. Before that plants were all we had throughout history to relieve pain and suffering. About 40 percent of the drugs we use today originated in plants, but they are now made with pure chemicals. That process is called synthesis.
Why have pharmacologists switched to synthesizing natural plants? Because plant roots can suck up heavy metals from the ground and thus contaminate the resulting drug. Plants can also add pesticides to the mix. Both medical marijuana and medical hemp do these things. Both plants can also grow mold, mildew, aspergillus, and other contaminants.
Chemicals are pure and pharmacologists do not have to worry about anything that can make a drug unsafe. Creating the drug with pure chemicals eliminates that concern.
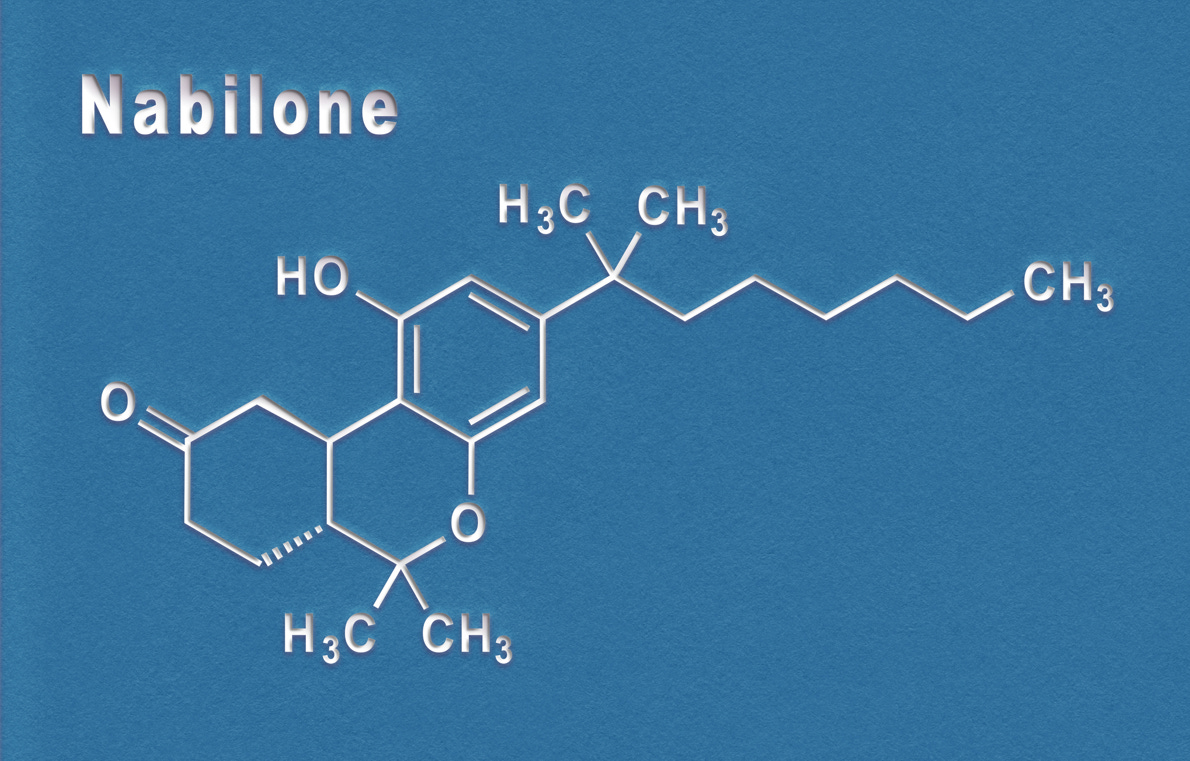
In 1985, FDA approved low-dose (2.5 mg, 5 mg, and 10 mg) Marinol (dronabinol) to treat nausea in chemotherapy patients who did not respond to standard medications. Seven years later, FDA also approved Marinol to treat wasting in HIV/AIDs patients. Cesamet (nabilone) was approved for both uses in 1985.
Cesamet was made by Ely Lilly but in 1989 the company pulled it from the market for commercial reasons. In 2006, another company, Valient, received FDA approval of Cesamet at low doses (1 mg to 2 mg twice a day; maximum dose 6 mg 3 times a day).
In 2016, FDA approved Syndros, an oral solution rather than pills, at low doses (5 mg/mL). However, it was later withdrawn from the market, again for commercial reasons.
All these drugs were made using pure chemicals.
Exceptions to the pure-chemicals rule occur with vaccines, some antibiotics, and in 2018 when FDA approved a purified version of CBD called Epidiolex for the treatment of three rare forms of seizures in children age one and above. Such seizures are so rare that most children who have them do not live to adulthood. “Purified” is a key word here.
FDA warns that purified CBD has many harms as well as unanswered questions revealed in clinical trials when FDA approved it for rare seizures. These include liver damage and toxicity in male animal studies and concerns about side effects when CBD is mixed with other medications.
If purified CBD could have such negative effects, what could raw CBD do to health?
Because the hemp industry found ways to add extremely high levels of THC to CBD, late last year Congress banned any CBD containing more than 0.4 mg of THC. The ban becomes effective in November of this year.
Nevertheless Dr. Memet Oz, head of the Center for Medicare and Medicaid (CMS), began giving FDA-unapproved raw CBD to Medicare patients on April 1, 2026. What’s more, FDA head Marty Makary says FDA will not interfere with the CMS plan.
Doctors can prescribe FDA-approved Marinol, Cesamet, and Epidiolex, and patients or their caregivers can buy them at pharmacies. Doctors cannot prescribe, but only recommend medical marijuana that states have “legalized” or medical hemp that Congress “legalized” until November 2026. Patients buy “legalized” cannabinoids found in marijuana and hemp at gas stations, convenience stores, vape shops, or dispensaries.
What kind of “medicine” is that? Why is FDA allowing this to happen?